| III. KERATIN DISSOLUTION |¶
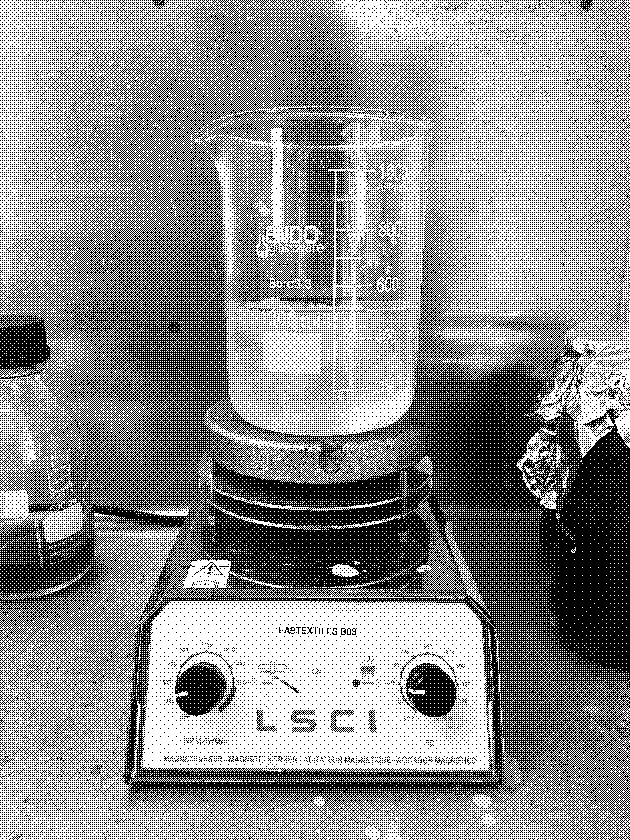
1. METHOD - SODIUM HYDROXIDE¶
What is SODIUM HYDROXIDE?

Ingredients¶
| a. | b. | c. |
|---|---|---|
| NaOH | Destilated water | Wool fleece |
| 60 g | 300 ml | 20 g |
| n. | Name | Price | Amount | Source |
|---|---|---|---|---|
| 01 | Sodium hydroxide (NaOH) | 4,10 € | 1 kg | Quimics Dalmau BCN |
| 02 | Distilled water | 3-5 € | 5 l | Fablab BCN |
| 03 | Local Merino wool | waste | - | Fablab BCN |
| 04 | Acetone | 8,47 € | 1 l | Quimics Dalmau BCN |
| TOTAL | - | 17,57 € | - | - |
Wool composite¶
| a. | b. |
|---|---|
| Acetone | Destilated water |
| 200 ml | 50 ml |
Tools¶
- 2x plastic bag (zipper), 2x measuring cups, spoon, 2x paper (coffee) filters - better to use folded lab filters, metal plate, magnetic stirrer (preheat function), dough roller, heat press, baking paper, paper roll, gloves, lab coat, protective glasses, gas respirator mask
PROCESS¶
Wool composite¶
Why we have to do it? We use acetone or ethanol to clean all dirty parts of wool fleece and remove first layers of wool fiber (cortex & cuticle). So we can more easily open the fiber bonds.
- Mix together 50 ml of distilled water and 200 ml of acetone.
- Put the wool inside of one of the plastic bags and pour acetone mixture inside of the plastic bag.
- Squeeze it by hand to distrubite acetone everywhere.
- Use a dough roller and press the plastic bag. Be careful not to pierce it. / You can also use a lab padder (3 times).
- Put it out of the plastic bag and remove the rest of water and acetone. Do not throw it away - we will use it later again.
- Preheat the heatpress. / You can use a lab pressure machine 2,2 - 3,3 bar.
- Place double sheets of baking paper on the surface of the heatpress and place there all pieces of wool with enough gaps between them.
- Heatpress it on 60°C for 20-30 min (more better).
- When they are pressed rinse them in the acetone again.
- Remove all the water and put them on an absorbent soft paper. Let it dry overnight.


Hydrolysis¶
- RATIO: 1 : 20 (water : NaOH)
- IMPORTANT: Please, be aware of the chemical properties of the chemical - NaOH. It is important to protect your skin - gloves, lab coat, protective glasses and do not forget gas respirator mask. Do not lean over the solution and keep your distance.
- Choose a safe place for your experiment. If it is possible do it outside or in a lab with proper ventilation.
- Pour in a borosilicate glass jar 300 ml of destillated water and weight 60 g of NaOH.
- Put NaOH into water. Never water into NaOH. And stir it with a spoon.
- Keep distance from the jar and do not lean over the solution. The solution will get warm, so do not be shocked.
- Place the jar with the solution on the magnetic stirrer and turn it on - RPM-Tr/Min 300 and 70°C.
- Cut the wool fleece in smaller pieces and put them to the jar for 24 h.





What will happen? The wool fiber will disappear it means that additional layers of wool fiber will be destroyed. You will achieve a liquid yellow/brown solution which you will need to precipitate. Because Keratin is now still mixed with NaOH.





Conslusion¶
| Timing | Disolubility | Biuret test | Difficulty | Safety |
|---|---|---|---|---|
| 24 h | Complete | Uncertain | Medium | High |
PRECIPITATION¶
Tools¶
- 3x measuring cups, spoon, 2x paper (coffee) filters - better to use folded lab filters, metal bowl, stirring spoon, weight, cooking pot (or kettle), cooking stove
Ingredients¶
| a. | b. | c. |
|---|---|---|
| Baking soda | Alum | Destilated water (hot) |
| 6 g | 18 ml | 200 ml |
Process¶
- Alum solution: Weight 18 g of alum and mix it with 100 ml of hot water. Add the alum into the water and mix them together, till the alum will disolve in the hot water completely. Keep stirring all the time.
- Add the alum solution into the glass with keratin extraction. Keep stirring all the time.
- Baking soda solution: Mix 6 g of baking soda with 100 ml of hot water. A reaction will appear, for that reason you need to place your plastic cup into another bigger bowl. While you stirring the keratin with alum solution add very slowly baking soda medium and wait for the reaction. In my case there was almost no reaction.
Result¶


Conslusion¶
Maybe next time I do not filter it and I will do the precipitation first. Also I will try another another amount of alum and baking soda.
REFERENCE¶
2. METHOD - UREA¶
What is UREA?

Tools¶
- magnetic stirrer, measuring cup, weight, spoon, kettle, paper filter (folded), gloves, lab coat, protective glasses, gas respirator mask
Ingredients¶
| a. | b. | c. | d. |
|---|---|---|---|
| Urea | M-bisulfite | NaOH | Distilled Water |
| 8 % | 0,5 % | 1 % | 90,5 % |
| 18 g | 1 g | 2 g | 180 ml |
| n. | Name | Price | Amount | Source |
|---|---|---|---|---|
| 01 | Urea | 9,50 € | 1 kg | Quimics Dalmau BCN |
| 02 | Distilled water | 3-5 € | 5 l | Fablab BCN |
| 03 | Local Merino wool | waste | - | Fablab BCN |
| 04 | Acetone | 8,47 € | 1 l | Quimics Dalmau BCN |
| 05 | M-bisulfite | x € | 1 kg | Quimics Dalmau BCN |
| TOTAL | - | x € | - | - |
Process¶
- Place a borosilicate glass jar on the magnetic heater and fill it with distilled water. Put the magnet inside and preheat it on 200'C.
- After it will get warmer put it on 60'C and turn on the stirring function. Slowly startt adding urea by spoon. Wait till it will disolve and add another spoon oof urea.
- When the all the urea is dissolved add M-bisulfate.
- Keep it mixing for a minute and finaly add NaOH.
- Before you will add wool try to cut it with scissors or shorten it by hands to make it short as possible.
- Be careful when you put it into the solution. Sometimes the fibers get stucked under the magnet on the bottom of the jar. In this case you have to control the speed.
- Let it stirring for 6 h on 3OO rpm and 60'C.
| Timing | Disolubility | Biuret test | Difficulty | Safety |
|---|---|---|---|---|
| 6 h | Incomplete | Positive | Medium | High |
Post-process¶
- The extracted solution was filtred through folded filtres and the filtrated solution was tested for protein using biuret reagent.
- The filtrated solution was then dialysised through a cow stomach (make sure to close all holes of the cow stomach) for 3 days - refill of distilled water every 3 h.
Conclusion¶
- The urea solutiion with M-bisulfate and NaOH was successful and almost no leftovers left aftter the extraction.
- Biuret test was positive in each case.
Last update: 2022-05-11