7. Textile as Scaffold#
Cristalization#
Experiments in Wemake#
Wednesday, during the wemake supplementary lesson we began to familiarize ourselves with the ingredients necessary for the realization of the crystals. Some solutions were simple to create for the availability of the elements (cooking salt, sugar) for others, however, as the borax were more complex to find.
I preferred to follow the process that Chiara Ganzerla realized and try to reproduce it at home to let the solutions sit but also to study them closely in their evolution.

Starting from right to left we find the solutions experimented: Cane sugar, Iodized salt, Sugar, Borax, Alum, Ionized Salt.
Unfortunately, the solutions have not had the opportunity to grow homogeneously or, as in the case of cane sugar, they even have a compact mass of crystal that has also imprisoned the strip of LEDs that we tried to cover with crystals.
Experiments at home#
At home I had only a few ingredients available and after numerous attempts to purchase in physical stores to get fast results I experimented with different types of salt and with Alum
Preparation#
I found an interesting gauze to help the crystals in their growth and together with a woolen thread I prepared the necessary jars and growth supports with the use of the scotch paper. To facilitate the realization of the various samples I dissolved the dye for alum directly in the jar with very little water.

for the preparation of the initial solution I followed the suggested proportion for the sugar crystal recipe 128g (1cup) with 87g (3 tablespoons). I later noticed that the solution became saturated using even only 40g of salt.

Let sit#
I let the compounds rest for more than 12h. already after a few hours the excess of salt and alum was deposited on the bottom.

Results#
Immediately the inks and dyes were decomposed when I poured the solution of Alum and water creating almost fatty lumps for the fluorescent ink and decomposing the liquid in the two pigments (red and blue) that I used to dye the solution. The bluest part has been integrated into the crystals while the red part has remained soluble in the water, tinging it decidedly red. The gauze instead reveals the intentional color: a pale purple.
Also in the case of blue we have a color decomposition that in this case becomes almost stringy. I imagine that the type of dye that in this case was for sweets, in powder, was the discriminant for having obtained a color in some cases not very homogeneous.
Fabricademy 6_growing salt and alum crystals from Greta Dalessandro on Vimeo.
Salt crystals#
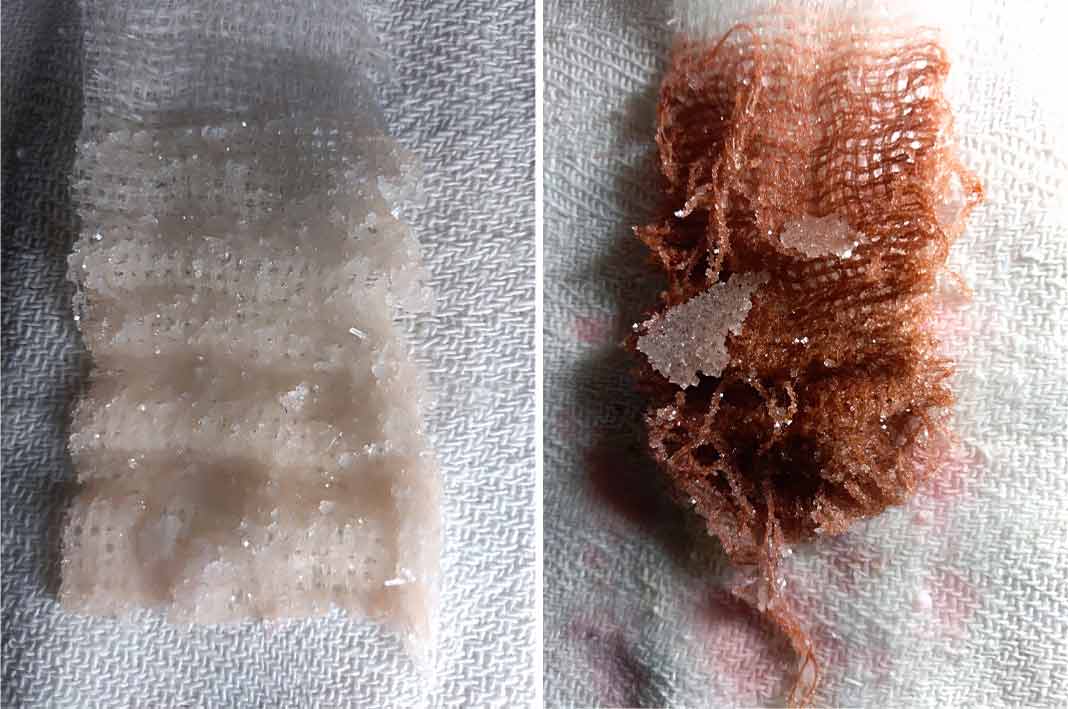
Clear Alum crystals#
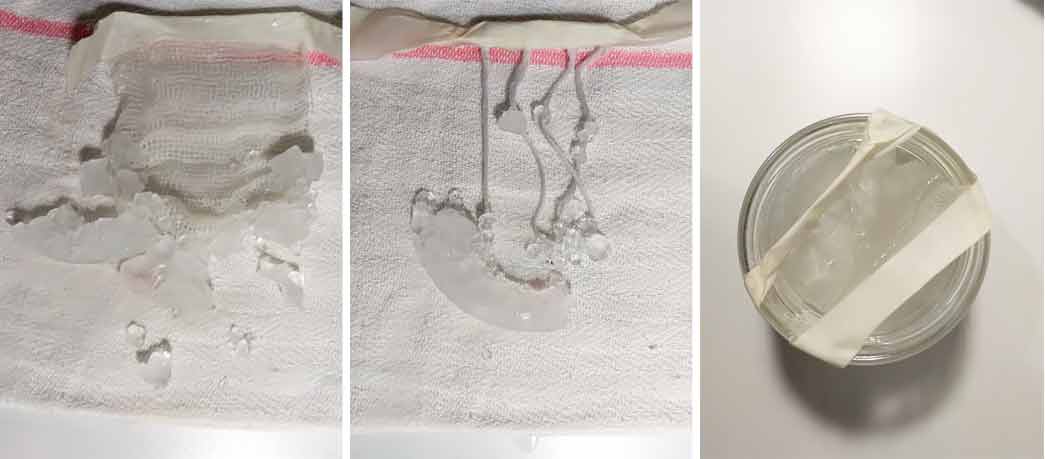
Purple Alum crystals#
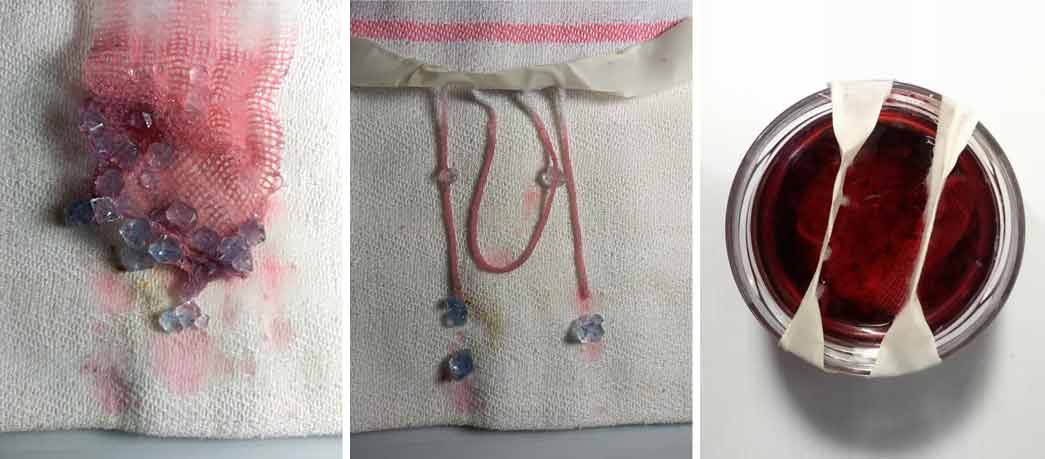
Blue Alum crystals#

Fluorescent Alum crystals#

Wood Textile#
I was so inspired that I thought about an entire collection of suitcases that I would like to develop (I do not know if inside the course or on my behalf). I found it really interesting to use a support as rigid as a canvas without elastene or a lycra as in my final choice. If possible I would like to work on the space that the individual modules have in such a way as to allow more freedom to the fabric or base on which the wooden modules are glued to expand. The structure remains rigid ase, does not collapse towards the inside but can expand slightly.
To actually test the flexibility of a wood textile I chose to create a simple, small bag to understand how the folds and the volume contained could change the structure of the shape.
On my Pinterest section I’ve selected some examples that inspired me to find a suitable pattern and a fairly small shape for this experiment.

I had at home a piece of thin enough wood (2mm) to work as a bag. I had previously applied a walnut veneer on the panel by veneering it.
This passage was made a few months ago and in some places the veneer had not adhered perfectly to the support. By laser cutting into small triangles, some layers were obviously divided.

Despite this inconvenience and with a good dose of patience I managed to get a first prototype pleasant and rather functional.

In a subsequent evolution and perfection of the project I would apply a hidden magnetic traingolar card that allows the bag to close as only the flap is closed at the moment.
7. Textile as Scaffold wood textile from Greta Dalessandro on Vimeo.