4. Biofabricating Dyes and Materials#
We had two very focussed lectures in a row this week, both from Cecilia - about the reasons why we’d use natural dyes instead of synthetic dyes (What an unhappy accident the discovery of synthetic dyes was) and about the range of sources for natural dyes, including bacteria. She also talked about bio plastics, giving examples of the alternatives and their uses. Our mission this week: to explore natural dyes and bioplastics and create samples of both. It’s been a busy few days.
plantbased dyes#
There are a huge range of examples of dyed fabrics as well as bioplastics and other natural materials here in the Material Library at the TextileLab Amsterdam. Below you can see some fish leather, bioplastics and various dyed materials.


things for the future#
preparation - weighing, scouring & mordants#
After looking at these, we began our own exploration by getting our materials ready - we were dying calico, cotton yarn and wool yarn, and a little bit of linen yarn. We made the yarns into little bundles by mimicking (somewhat ineptly on my part) Cecelia’s grandmother’s technique, and ripped the calico into strips. We weighed everything while it was dry (so we know how much dye material we need) and divided it up into bundles so that we could do three kinds of mordant - alum, copper and iron - for seven kinds of dye. There was maths involved, and I nodded supportively while Cecilia did that out loud.
For your benefit, I’m including my notes on how to make bundles of yarn like Cecilia’s grandmother - how you interpret these, is up to you. Obviously, mine was not video-d, which I think is a good thing for all of us.

We scoured everything except the wool in simmering hot water with carbonate of soda, to clean it of anything that will inhibit the dye. Then we rinsed it and submerged the materials in the different mordanting solutions. Each of these has a different effect on the quality of the colour you achieve when you submerge the materials in the dye bath. You can see below our piles of plain materials and then the same materials after we mordanted them (the ones that were in the copper bath are the greenish ones, the others are visually unchanged) and then a photo of the same materials dyed.



We had about 10 strips of calico and 25 ‘hanks’ of yarn for each dye batch, with approximately equal numbers of the different mordanted materials.
the dye bath#
I was the keeper of the campeche pot because I expressed interest in playing with a material that isn’t available in New Zealand. Campeche is a bark from South America. Feeling great responsibility, I weighed out the amount of plant fibre according to the instructions (half the weight of the material we were dyeing) and started simmering it in about 5 litres of water. You will see below the jars full of the different dyes (madder, turmeric, annatto, hibisus etc) and on the right the amazing colour of the water when I added the campeche.


the outcome#
You can see below the range of colour I got from the different materials with their different mordants, which ranged from a medium blue in calico (alum mordant and then dipped in vinegar to modify the pH afterwards) through to almost black with the wool that was mordanted with iron (no PH modifier). It was hard to see any effect from the alum and the copper mordants, especially on the calico, but once I’d dipped them in pH modifiers (soda and vinegar) they were different, as you can see on the right.


alcohol & PH playtime#
While we were waiting for the dye baths to be done, we played around with extracting colour with 94% alcohol… and with adjusting the colours with PH as well. It was like magic when they changed colour. We used carbonate of soda or vinegar to do this.


Understanding the amount of time, chemistry and water involved in dyeing fabrics was very interesting, and made the bacteria dyes even more appealing.
bacteria dyes#
A technique developed and tested here at the TextileLab, bacteria is used to stain fabric. We were introduced to the WetLab and the protocols we needed to follow to maintain a clean space. The bacteria will be used to dye the fabric in two ways - by encouraging the bacteria to grow along the fabric with a petrie dish, and by extracting the pigment from dead bacteria to dye material.
preparation is everything#
Over the last three years in our DIYbio Lab at Fab lab Wgtn we have learnt about clean processes because we’ve been growing mushrooms. It was nice to see that the protocols are similar, and I look forward to doing more in this WetLab here.
We put labcoats on, tied our hair back and constantly washed our hands, gloves and useful surfaces with alcohol. There was also a ‘bunsen’ burner involved for sterilising our tools, which were tweezers and a scraper.
the growing media#
We had two jars of growing media - an agar, jelly-like mix called nutrient agar and a liquid mix called LB broth, both delicious food for our bacteria. We prepared a plate of each media, after these had been boiled in a pressure cooker to kill any contamination. You can see below the two jars with heat sensitive autoclave tape over the lids. The tape develops the dark stripes when it has been heated enough so that we can tell that we’ve succeeded in creating the temperatures needed to sterilise. We also sterilised our silk fabric squares (which we had manipulated in various ways) in the cooker in a plastic bag that won’t melt in the heat.


the bubble of sterile goodness#
A combination of alcohol evaporating from the table surface and the bunsen burner burning creates a sterile bubble for us to work in while we’re transferring the solutions into individual petrie dishes. To maintain the bubble we don’t talk, breathe shallowly (or not at all) and work quickly.
plating & scraping & incubating#
Cecilia showed us the workflow we needed to follow to plate up our bacteria. First of all we labelled each dish with the date, the media and our name. The images show me scraping the bacteria plate with the sterilised scraper, trying to lift the lid as little as possible, and applying it to the liquid media and my fabric square. The idea is that the media soaks into the fabric and the bacteria follows it. Hopefully my beautiful knots create pathways and roadblocks that will end up with the bacteria making interesting patterns on my silk.
I also poured the agar mix into my other petrie dish and when it had cooled and set, scraped some bacteria onto that. This will be the plate that we extract the pigment from (which is apparently bacteria poohs).


The image below is of my silk in the petrie dish. I hope to be able to add a photo next to it, in a week or so, that shows the bacterial results. Fingers crossed…

Once we’d all gone through this process, Cecilia wrapped the dishes in groups of two or three with a stretchy tape to keep them in a sterile bubble, and then they went into the incubator where they’ll be sitting at a constant 24 degrees until Monday or Tuesday.

the big reveal#
So, Monday arrives, and we’re all very excited to see how our bacteria have grown. We went en masse to the WetLab and proceeded to remove our petrie fabrics from the incubator and go through the process of killing the bacteria. You can see in the image below that gloves and lab coats were donned and you will be able to imagine that we washed our gloves with an alcohol solution again as well as keeping the door to the lab closed so that no nasties could waft into the incubator while we were doing this.
Cecilia had shown Jessica the process for pressure cooking the life out of the bacteria, and she had noted them down, so we followed the instructions.


…weeeell, in all honesty, there were so many of us in the room that I left after a while. I was obsessing about what I’d been working on, so when I realised that five of us would be watching a pot boil, I decided to leave. When everything contaminatable (I’m sure that’s a real word, but I don’t think I’ve spelt it correctly) was closed I slipped out of the room and went back to doing that. Which was re-drawing my Circular Fashion project so that it is ~parametric.
What seemed only minutes later but actually an hour later… I received the message below and raced upstairs. Which is a fairly risky business, as you might guess from the image below of Bella cautiously posing on the stairs.


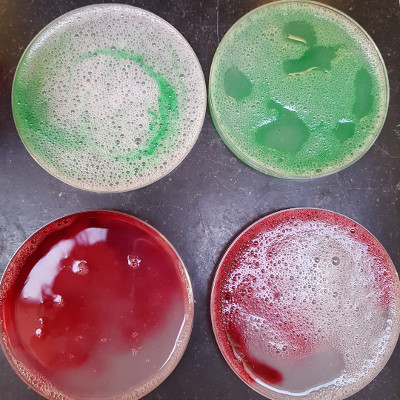
Anyway, my tiny knotmaking paid off, as I got an awesome result from the bacteria dye. Also, our attention to sterile protocols paid off as well, otherwise it would have been deeply disapointing. I’m so pleased, that I’m allocating the full 900px of real estate on this page to two photos… It’s so beeeeeaaauuuuttiiiifuuuulllll…

…and here it is hanging on our washing line in the TextileLab. The bit I didn’t understand is how tying a round knot can make squares and rectangles, but apparently it’s because of the linear nature of the warp and weft of the fabric.

Between making the plates and killing the bacteria, we made some…
bio-plastics!#
We made a couple of batches of the standard gelatin recipe in the wiki, and added very cheap liquid soap to one so that we could make two kinds of plastic - one like a foam and the other just a plain sheet.
generic tests#
We then added our own colours and moulded our tests in different ways. Images of my samples are below, with thanks to Jessica for the action shot of me pouring gelatin into the pot.
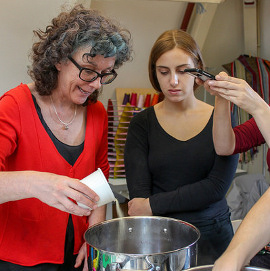


It was fun playing with the childhood-system-that-shall-not-be-named-in-case-copyright-is-breached as a mould - just putting some gaffer tape around to create a box for the jelly to sit and set.



And, you’ll remember that I loved visitng Mycropia the other week - well, I thought that this sample showed a lot of promise, with the way the dye was distributing, but it went disapointingly two-dimensional when it set. However, it started to also grow mould over the weekend. Interesting times…


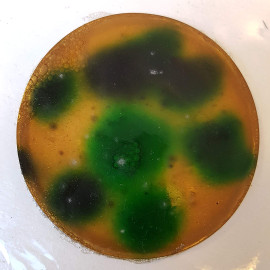
double double toil and trouble#
Anyway, after this, I decided to play with my own recipe. I have done quite a bit of bio-plastic experimentation at Fab Lab Wgtn, mostly with agar (but sometimes with guar gum, tapioca and other vege-based polymers), and I have had some good results there.
While I was living in Dubai and working at Fab Lab UAE this year, I bought some gum arabic at a market. Eng. Hashim, who I worked with at the time, helped me to understand what I was buying, because it was new to me to see these things in this way. The gum is at the back of the image below, behind the turmeric.

This is an image of some of the people I worked with at Fab Lab UAE. The image is taken by Francisco who is 4WDriving across Spain at the moment. If you can work out who I am, you will see that Hashim is next to me on one side and Francisco is on the other side. Here’s another clue: Hashim is the other stylish-looking person wearing glasses.

ANYWAY, when I left Dubai, I took the gum arabic with me to the Fab14 conference and because I knew I couldn’t bring it to New Zealand due to all our bio-security laws, I ‘gave’ it to Cecilia. Ten weeks later, and we’re doing this project - I casually ask, with fingers crossed: Do you still have that gum arabic… and of course, Cecilia smiles, opens a drawer, and pulls out the unopened packet, saved for me.
With absolutely no idea how this is going to behave - is it a polymer or an elasticiser? - I decide that it’s an elasticiser and that I should combine it with agar and, in the first instance, without glycerol.



I chose a small lump and mixed and heated and stirred and heated and scraped and heated and then mixed and heated and stirred and heated and scraped and heated some more, by which time the water reduced to the point where it had halved and was looking very small in the giant pot, so I gave up and took what was left of the lump out and weighed it.
the recipe#
From the original 13gm it was now 6gm, so the recipe is:
Add
- 7g gum arabic to
- 260ml water
- simmer until water is reduced to half while melting lump of gum arabic
- add 3gm agar and simmer until completely dissolved.
- pour into 2 x petrie dishes, lids and bases.
- colour as you wish
- anxiously leave on sunny windowsill over the weekend as you’ve had the experience of mould growing if it doesn’t dry out fast enough (it is agar, after all)
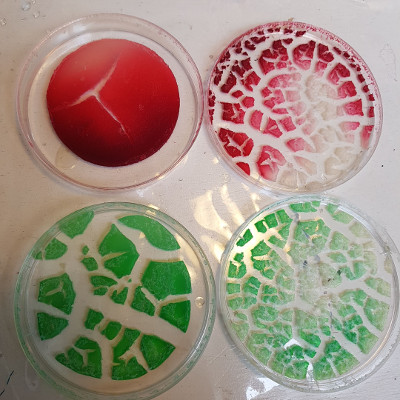
the outcome#
Which meant that it all dried and cracked. This was the only one that still looked promising, so we’ll see how that progresses in the coming days. Sigh At least I still have the dye successes.
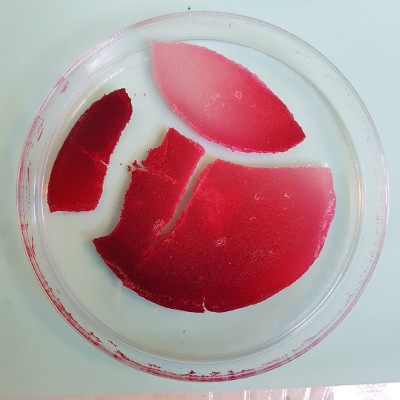
During the global review, Anastasia suggested that I pour a new batch of bio-plastic over the original one - an awesome idea, so I did it. This time I made the recipe with:
- 14g gum arabic
- 18g agar
and I dissolved these in a jar of
- 200ml boiling water
I left this overnight and then ‘double-boiled’ it. You can see in the photo below what that means - I put the jar in a pot of boiling water for 15 minutes.

This helped to finish dissolving the two materials, and then I added
- 4g glycerine/glycerol
- 200ml water
And put it all directly into the pot. I simmered it all for a further 15 minutes, then poured it over the original bioplastic samples. The colour in the existing pieces started to bleed quite beautifully into the new solution. You will see also, when I removed the samples from the petrie dishes and turned them over, that the original samples had rehydrated and they looked like reptilian skin.



The photos below show that the bioplastic has gone from being similar in texture to the gelatin, to now being almost foamy. The water seems to have been semi-trapped in bubbles of the solution, which weren’t at all visible until the water made its way out. It’s physically light and squeezable. I could make a pressure sensor out of it…


bonus section#
Dutch Design Week. I’m just going to add images - it was fun and inspiring and nice to see everything and everyone.






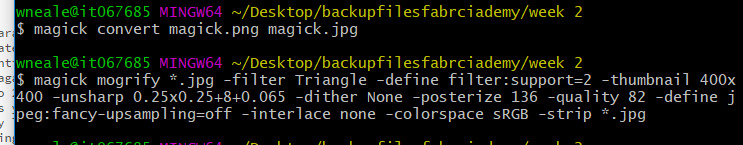
image resizing with image magick - I found a post with code that I messed with which was fairly effective but I’m still mostly sticking with gimp. The #ddw photos (jpg only) have been resized using this and the code is as follows:
magick mogrify *.jpg -filter Triangle -define filter:support=2 -thumbnail 400x400 -unsharp 0.25x0.25+8+0.065 -dither None -posterize 136 -quality 82 -define jpeg:fancy-upsampling=off -interlace none -colorspace sRGB -strip *.jpg
This is explained in lots of detail in the post, but basically it resizes and compresses your images in a batch with one copy-paste-enter in terminal. Unfortunately it also rotates more than half of my images, and I can’t work out why, which is why I’m sticking to gimp still.
to be continued…